Traditionally, little attention has been given to fine details in hair restoration, especially in reconstructive surgery. The techniques used most frequently in the past include tissue expansion, scalp flaps, hair plugs, and scalp strips.1-7 Some of these techniques are still used in hair restoration, especially tissue expansion and flaps.
The introduction of micrografts (1 to 2 hair grafts) and mingrafts (3 to 4 hair grafts) was a significant advancement in the treatment of male pattern baldness and female androgenic alopecia. Finally, natural and aesthetically pleasing results were possible.8-11.
Subsequently, micrografting and minigrafting were used to correct unfavorable results from previous hair transplantation,12,13 sideburns lost following rhytidectomy,14 and burn alopecia.15 In addition to the use of these grafts to treat male pattern baldness and female androgenic alopecia, many other applications have been found in the aesthetic reconstruction of the face and scalp. In my experience, micrografts and minigrafts have been used in aesthetic reconstruction of the face and scalp in the following order of frequency:
1. To restore hair lost as a result of (iatrogenic) postsurgical causes (i.e., after facial rejuvenation procedures or procedures involving incisions on hair-bearing facial skin or scalp; Figs. 1 and 2).
2. To correct unfavorable results from previous hair transplantation, to eliminate the “plug look” caused by the old hair plugs technique, and to camouflage scars from flap procedures or scalp reductions (Fig. 3).
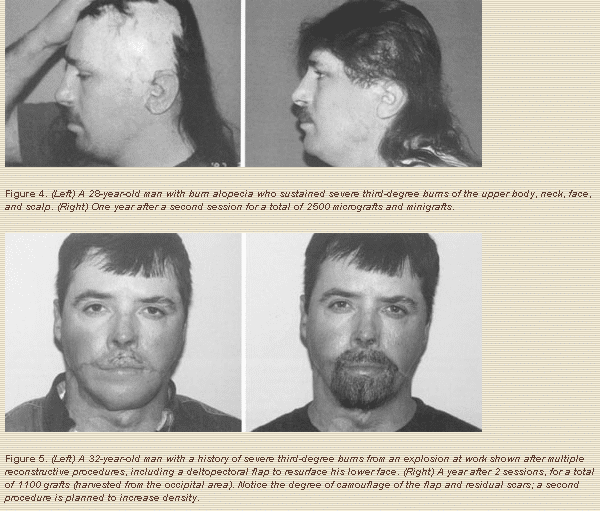
3. To restore scalp and facial hair lost as result of burns or traumatic injuries (Figs. 4-6).
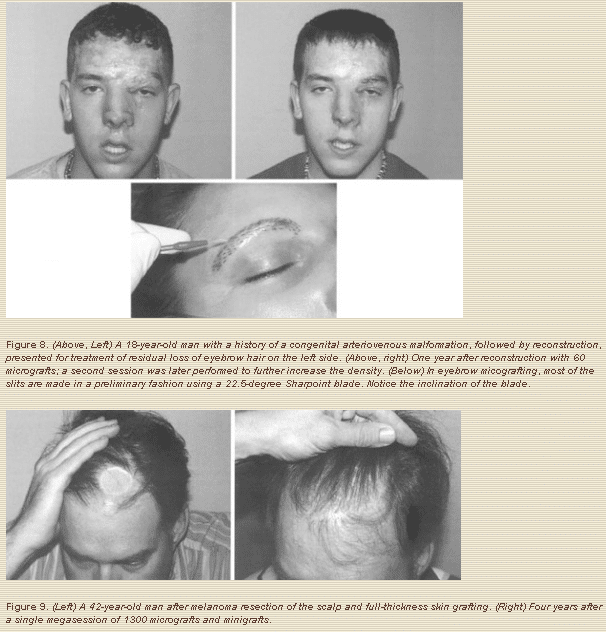
4. To restore hair loss attributable to congenital reasons, such as complete bilateral cleft lips (no mustache hair in the prolabium) and triangular alopecia, or to remove congenital nevi or arteriovenous malformations (Figs. 7-8).
5. To restore hair loss after oncologic resections (i.e., resection of melanoma and other skin malignancies) (Fig. 9)

Technique
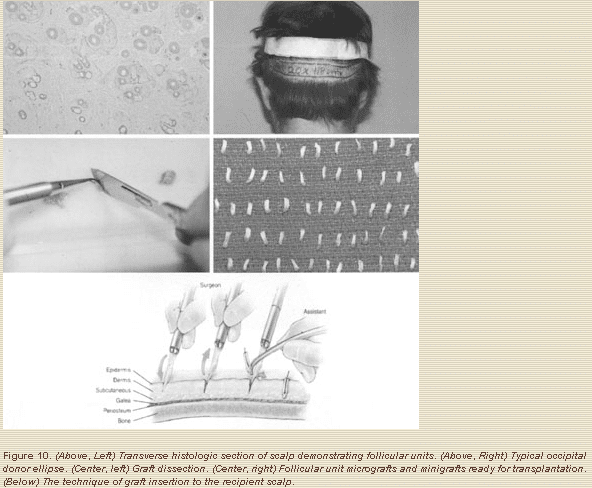
Usually, these procedures are performed with the patient under intravenous sedation and local anesthesia, unless the patient is a child, in which case the procedure is performed under general anesthesia. Midazolam (Bedford Laboratories, Bedford, Ohio), usually 2 to 10 mg, and fentanyl (Abbott Laboratories, North Chicago, Ill.), usually 50 ug, are used for the sedation; 0.5% bupivacaine (Abbott Laboratories) with epinephrine 1:200,000 is used for nerve blocks and local infiltration. The technique for scalp hair transplantation is basically the same as the one described previously as slit and immediate insertion of the grafts.8-11 With the patient in the supine position and mildly sedated, occipital nerve blocks and supraobital nerve blocks are created with 0.5% bupivacaine with epinephrine 1:200,000; the donor site is harvested from the occipital area as a horizontal ellipse (Fig. 10, above, right). Subsequently, a mild tumescent infusion is given using 0.25% lidocaine (Xylocaine; AstraZeneca LP, Wilmington, Del.) with epinephrine 1:200,000 (approximately 30 cc) for hemostasis, in order to intentionally produce temporary edema, which facilitates graft insertion.
With a no.10 Bard-Parker blade, the incisions for harvesting the donor ellipse are made parallel to the hair shafts using 3.5x loupe magnification, then 2-mm-thick slices are dissected from the donor ellipse. The actual grafts are dissected from these slices by technicians, who make an effort to maintain the follicular units intact under 3.5x to 10x magnification (Fig. 10, above, left and right, and center left and right).
For the front 5 to 10 mm of the scalp, a 22.5 degree Sharpoint blade is used; the surgeon makes a slit, and an assistant with a fine curved jeweler’s forceps immediately inserts a graft into the slit (Fig. 10, below). Because these blades are so small and sharp, they leave almost no detectable scar. Further posteriorly, a no. 11 Feather blade is used. Slits made with the no. 11 Feather blade heal beautifully but occasionally may leave a slightly detectable scar (minimal pitting or pigment changes); therefore, it is best not to use this blade in the front hair line.
Hair restoration of the face (eyebrows, mustache, and sideburns) is more difficult because as one makes slits near other grafts, the neighboring grafts tend to “pop out” of their corresponding slits. On the face, slits are made in a preliminary fashion, with the grafts inserted a few minutes later. Also, a 22.5-degree Sharpoint blade is used. Sometimes it is helpful to use an 18-guage NoKor needle (Becton Dickinson & Co., Rutherford, N.J.) at the time the graft is inserted because it will act as a sort of dilator; then, again with a jeweler’s forceps, an assistant introduces the graft as the NoKor needle is withdrawn.
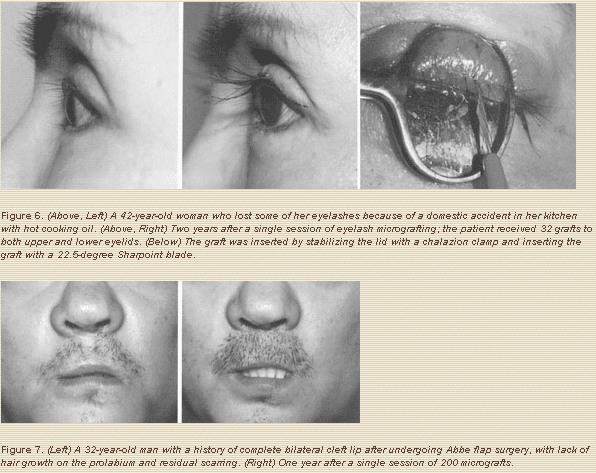
Close attention should always be paid to the natural direction of hair growth; the blades and Nokor needle should be inserted in the desired direction of hair growth. For the eyebrows, the blade is placed at a very acute angle up and laterally, in an attempt to follow the natural direction of eyebrow growth (Fig. 8). For the mustache, the blade enters in a caudal to cephalic direction and as flat as possible to the lip surface (10 to 15 degrees) to promote a downward growth. The same principles apply to beard restoration.
On the eyelashes, the technique is a bit more complicated because the eyelids are thin, delicate, more mobile, and intimately adjacent to the eye globe. To insert follicular unit micrografts into the eyelid margin, a 15-degree or a 22.5-degree Sharpoint blade is used, with immediate insertion of the grafts. A chalazion clamp is used to stabilize the lid margin and protect the eye globe. Often, 10 or 12 one-or two-hair micrografts are inserted per upper eyelid; more than one session may be required for an optimal result. These sessions are performed 8 to 12 months apart.
If the donor source is the scalp, as is often the case, the patient most be told in advance that the new lashes will grow at the normal scalp hair growth rate, so periodic (perhaps weekly) trimming will be needed. The most difficult aspect of eyelash restoration is controlling the direction of hair growth. For the upper lids, the hair is aimed out and down. In my experience, it is easier to control the lashes of the upper lids (straight forward) than those of the lower, especially because one does not want the lower eyelashes to touch the cornea (Fig. 6). Eyelash curlers may be beneficial to achieve the desired curvature. Nasal vibrissae are also reasonable potential donors.16 Eyebrow hair has more similarities to eyelashes than scalp hair with regard to texture and speed and length of growth (8 to 12 mm); therefore, when feasible and available, the eyebrows are certainly a favorable donor site.
All grafts should be inserted to a depth that leaves the epidermis of the graft superficial to the epidermis of the recipient site; in this fashion, ingrown hairs and inclusion cysts are prevented. This applies to all areas of hair transplantation. At the end of the procedure, the grafts should look like little bumps; as the healing takes place, the epidermis of the grafts becomes a crust and finally sheds after approximately 10 to 14 days, leaving a smooth area. It takes a minimum of 3 to 4 months before any hair growth is seen; growth then continues to improve over the next few months until the final result is evident at approximately 10 to 12 months.
Results
From 1993 to 2001, I performed aesthetic and reconstructive hair transplantation in more than 500 cases. Every graft was inserted by me personally. The majority of these cases (90 percent) were performed for purely aesthetic reasons to treat male pattern baldness; the remaining 10 percent were reconstructive cases.
In this series of 500 cases, there were only two cases of keloid scarring at the donor site (0.4 percent); fortunately the scarring was not at all recipient area. Two cases of poor hair growth (0.4 percent) occurred after the procedure; this happened early on, when the assistants were learning to cut grafts. Two cases of donor-site postoperative hair loss occurred around the scar, most likely attributable to tightness of the closure (0.4 percent). The total complication rate was 1.2 percent. In patients with realistic expectations, the satisfaction rate was extremely high, more than 95 percent after one session. Patients who were not satisfied after one session often became satisfied after a second session.
The grafts must be dissected carefully and atraumatically, and they must be maintained wet with normal saline throughout the procedure or no hair will grow. During long procedures (more than 3 to 4 hours), it is also important to keep the grafts chilled on normal saline, resting over a basin filled with ice.
Discussion
Because of their small size, micrografts and minigrafts appear to have a lower metabolic requirements to survive. They tend to grow in areas of fibrosis and burn scars and over skin grafts and flaps, including split-thickness skin grafts. The rate of survival and ultimate hair growth under these circumstances appears to be approximately 85 percent, compared with approximately 95 percent on unscarred, healthy tissue. More recently, follicular unit grafts have been used in hair transplantation. For eyelash reconstruction, use of a 23-guage, curved needle has been described in the literature. The graft is fed into position using the eye of the needle, with the long hair of a single hair graft (acting as the thread) entering the eyelid some 8 to 10 mm cephalic to the ciliary border of the eyelid and emerging at the free margin of the eyelid.17
Another interesting and recently described technique that I am evaluating is the use of a scalp strip inserted along the ciliary border of the eyelid. A pocket is made by evaluating a skin flap superficial to the orbicularis oculi muscle for the upper lids; a (hair) scalp strip is placed into the pocket. This technique can give quite a bit of hair density in a single session.18
The “follicular unit” concept was originally described by Headington in 1984.19 He studied the transverse histologic anatomy of the scalp and found that hair grows in groups of one, two, three, or four hairs, with independent sebaceous glands, sweat glands, neurovascular bundles, and piloerectile muscles surrounded by a sheath of collagen. These are true physiologic units, and an effort must be made to keep them intact, because doing so appears to increase graft survival and ultimate hair growth. Of the various techniques described in the past for hair restoration in reconstructive cases of the face and scalp, there definitely remains a place for tissue expansion and, in selected cases, scalp flaps and free-tissue transfers.
When feasible, I prefer to use follicular micrografts and minigrafts for the following reasons: the procedure is safe; sedation and local anesthesia can usually be used; there are no worries about potential exposure of tissue expanders or flap ischemia or necrosis; often, reasonably good results can be obtained in a single session (although a second session is sometimes needed to increase hair density); the scars are undetectable; and there is minimal down time and no need for a hospital stay, as this is an ambulatory procedure that can usually be performed in the office surgical suite.
The main disadvantage of this technique is that it is a tedious process for which a great deal of patience is required from both the surgeon and the assistants. There is, of course, a learning curve, especially in learning to pack the grafts close enough to one another to obtain a reasonable degree of density in just one or two sessions; however, it can be done. As with all other surgical procedures, the more we perform them, the more efficient we become and the more our results improve.
Conclusions
The use of follicular unit micrografts and minigrafts for reconstructive purposes on the face and scalp has been found to be safe and predictable and to provide aesthetically pleasing results. There is a high level of patient satisfaction, even after a single session.
References
1. Leighton, W., Johnson, M., and Friedland, J. Use of the temporary soft-tissue expander in postraumatic alopecia. Plast. Reconstr. Surg. 77: 737, 1986.
2. Buhrer, D. P., Huang, T. T., Yee, W., and Blackwell, S. J. Treatment of burn alopecia with tissue expansion in children. Plast. Reconstr. Surg. 81: 512, 1988.
3. Hudson, D. A., and Grobbelaar, A. O., The use of tissue expansion in children with burns of the head and neck. Burns. 21: 209, 1995.
4. Cronin, T. D., Use of hair-bearing punch grafts for partial traumatic losses of the scalp. Plast. Reconstr. Surg. 42: 446, 1968.
5. Giraldo, F., Gonzalez, C., Garnica, I., Ferron, M., and Rus, J. A. Sideburn reconstruction with and expanded supraauricular trapezoid flap. Plast. Reconstr. Surg. 100: 257, 1997.
6. Kasai, K. I., Ogawa, Y., and Takeuchi, E. A case of sideburn reconstruction using a temporoparietal-occipital island. Plast. Reconstr. Surg. 87: 146, 1991.
7. Harii, K., Ohmori, K., Ohmori, S. Hair transplantation with free scalp flaps. Plast. Reconstr. Surg. 53: 410, 1974.
8. Uebel, C. O. Micro and minigrafts: A new approach to baldness surgery. Ann. Plast. Surg. 27: 476, 1991.
9. Barrera, A. Micrograft and minigraft megasession hair transplantation: Review of 100 consecutive cases Aesthetic Surg. J. 17: 165, 1997.
10. Barrera, A. Micrograft and minigraft megasession hair transplantation after a single session. Plast. Reconstr. Surg. 100: 1524, 1997.
11. Barrera, A. Refinements in hair transplantation: Micro and minigraft megasession. Perspect. Plast. Surg. 53: 410, 1974.
Alfonso Barrera, M.D.
West Houston Plastic Surgery Clinic, P.A.
915 Gessner Rd., Suite 825
Houston, TX 77024
